Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Okubo, Takahiro*; Yamazaki, Akio*; Fukatsu, Yuta; Tachi, Yukio
Microporous and Mesoporous Materials, 313, p.110841_1 - 110841_11, 2021/01
Times Cited Count:6 Percentile:35.32(Chemistry, Applied)Pore distributions in water-saturated Ca-montmorillonite were investigated using  H NMR measurements under various dry densities (0.8 - 1.6 g/cm
H NMR measurements under various dry densities (0.8 - 1.6 g/cm ) and porewater salinity conditions (deionized water, 0.1 and 1 M CaCl
) and porewater salinity conditions (deionized water, 0.1 and 1 M CaCl ), at the temperature range of 233 - 303 K. The volume fractions of the interlayer pore including two and three hydrated layers and the non-interlayer pore in compacted Ca-montmorillonite were quantified by NMR relaxometry including
), at the temperature range of 233 - 303 K. The volume fractions of the interlayer pore including two and three hydrated layers and the non-interlayer pore in compacted Ca-montmorillonite were quantified by NMR relaxometry including 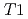 and
and 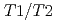 distribution analysis, and were compared with NMR cryoporometry and X-ray diffractometry. These analysis provided consistent pictures on the pore distributions in compacted Ca-montmorillonite, in contrast to Na-montmorillonite. The main factor affecting the pore distribution in compacted Ca- and Na-montmorillonite is the density, whereas the effect of porewater salinity is relatively smaller. The effect of interlayer cations is also relatively smaller at higher density, although the differences in the pore structures are significant at low density.
distribution analysis, and were compared with NMR cryoporometry and X-ray diffractometry. These analysis provided consistent pictures on the pore distributions in compacted Ca-montmorillonite, in contrast to Na-montmorillonite. The main factor affecting the pore distribution in compacted Ca- and Na-montmorillonite is the density, whereas the effect of porewater salinity is relatively smaller. The effect of interlayer cations is also relatively smaller at higher density, although the differences in the pore structures are significant at low density.
Awual, M. R.; Hasan, M. M.*; Ihara, Toshihiro*; Yaita, Tsuyoshi
Microporous and Mesoporous Materials, 197, p.331 - 338, 2014/10
Times Cited Count:182 Percentile:99.2(Chemistry, Applied)Awual, M. R.; Hasan, M. M.*
Microporous and Mesoporous Materials, 196, p.261 - 269, 2014/09
Times Cited Count:223 Percentile:99.57(Chemistry, Applied)El-Safty, S. A.*; Shenashen, M. A.*; Ismael, M.*; Khairy, M.*; Awual, M. R.
Microporous and Mesoporous Materials, 166, p.195 - 205, 2013/01
Times Cited Count:137 Percentile:98.08(Chemistry, Applied)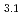 (OCOCH
(OCOCH )
)
 0.9H
0.9H O
OKozai, Naofumi; Mitamura, Hisayoshi; Fukuyama, Hiroyasu; Esaka, Fumitaka; Komarneni, S.*
Microporous and Mesoporous Materials, 89(1-3), p.123 - 131, 2006/03
Times Cited Count:11 Percentile:39.24(Chemistry, Applied)Layered transition metal hydroxide salt (LTMHS) is a group of anion-exchangeable layered compounds. Although LTMHSs have recentely attracted attention of researches on anion exchange and intercalation, very limited numbers of reports have been published on their synthesis, characteristics, and applications. This paper describes basic characteristics of a new LTMHS, nickel-copper hydroxide acetate. Hydrothermal Heating of an aqueous solution containing nickel acetate, copper acetate, and hydrogen peroxide to 150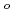 C for 4h yielded a layered compound with an analytical composition of NiCu(OH)
C for 4h yielded a layered compound with an analytical composition of NiCu(OH)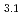 (OCOCH
(OCOCH )
) 0.9H
0.9H O. This compound does not take up Cl
O. This compound does not take up Cl and NO
and NO
 in aqueous solution but takes up multivalent anions and shows high selectivity in uptake of toxic SeO
in aqueous solution but takes up multivalent anions and shows high selectivity in uptake of toxic SeO
 and AsO
and AsO
 . This compound may find applicarion in the removal of those toxic anions form natural water and wastewater rich in Cl
. This compound may find applicarion in the removal of those toxic anions form natural water and wastewater rich in Cl and NO
and NO
 .
.